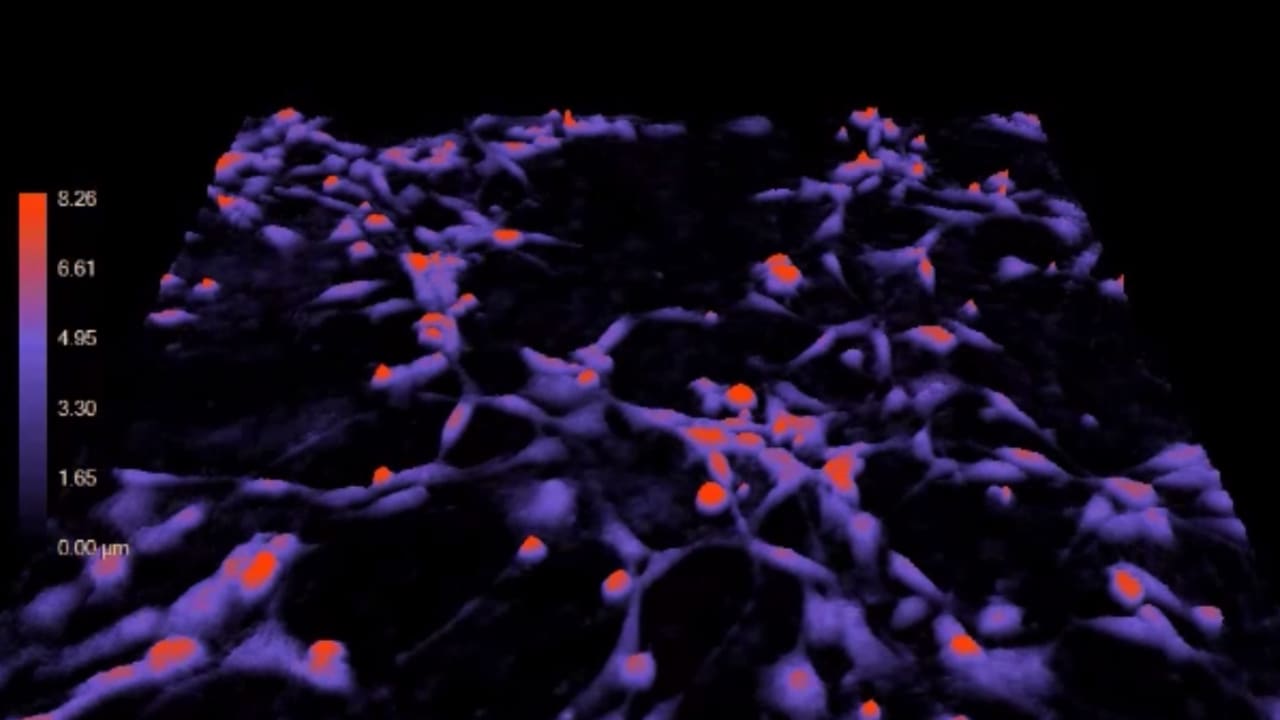
Researchers at University of Virginia were able to change the course of stem cells, making them turn into brain cells instead of becoming heart cells-- simply by turning off a single gene.
The findings, peer-reviewed in the journal Nature, could help scientists understand how specific genes affect the development of the human body and the role they play in developmental diseases, potentially leading to new therapies.
“Previously, it's been thought that the paths that cells take towards becoming a heart cell or a nerve cell are very rigid,” said researcher Jeff Saucerman, PhD, of UVA’s Department of Biomedical Engineering. “This study is showing that this process is actually much more fluid.”
The research, which used CRISPR genome-editing approaches to turn off the Brm gene in mouse stem cells undergoing canalization into heart cells, resulted in the mouse cells lacking a protein called Brahma. It challenges fundamental ideas about how stem cells progress to mature body cells and notes that stem cells should be seen as a "blank slate." It's the first to explore Braha's impact on cardiac differentiation.
Researcher Ali Khalilimeybodi, who developed the computer model used to guide the research, noted the unconventional approach used. "By using the computational models, we get a deeper understanding of the mechanism that Brahma can coerce changes in the cell fate, or differentiation, process.”
Added Saucerman: “There's an awful lot to be explored on what happens after – how do these cells become mature, highly contractile cells? I think that's a major challenge for the field,” Saucerman said. “That has therapeutic implications, because we need to be able to develop mature cells to then transplant into patients or to discover new drugs.”