Around 37 million Americans and 800 million people worldwide suffer from chronic kidney disease (CKD), according to the American Kidney Fund and research published by the University of Tennessee.
Despite more than a million people dying from CKD a year, few advancements in treatment have developed in more than a decade. Pharmacological therapy to delay CKD remains elusive. And the only actual treatment solution is dialysis, which dramatically reduces the quality of life for CKD patients.
Until now.
Curespec was founded in 2020, and its first market-ready product, Nephrospec, shows promise in treating CKD by promoting new blood vessel formation and restoring damaged tissue function using its patented shockwave technology.
eHAT electro-Hydraulic Acoustic Therapy (eHAT) is an acoustic wave treatment with much less intensity than traditional shockwave treatment, which could result in kidney damage.
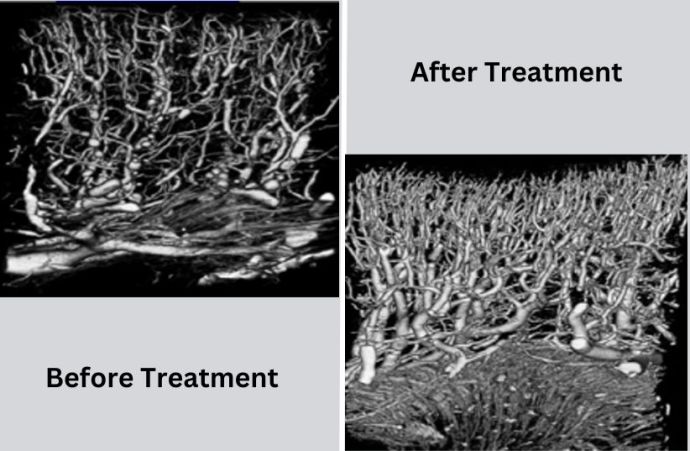
In several animal studies, eHAT has improved healing in tissues, such as bone, muscle, heart muscles and kidney, mainly via stimulating somatosensory receptors and upregulating vascular endothelial growth factors, easing inflammation and fibrosis, ultimately healing kidney tissue, preserving kidney function and delaying CKD progression. It can also attract stem cells into injured kidneys for tissue repair.
eHAT has been used for other indications for almost two decades, including some by Curespec's sister company Medispec. For example, several studies have shown eHAT effective for treating musculoskeletal disorders, myocardial ischemia and erectile dysfunction.
Curespec was founded by Avner Spector, who has worked in the medical device field since the 1980s. His parents founded the technology company Spectronix. Spector has a degree in mechanical engineering. However, he became passionate about being an inventor of medical equipment and decided to try it. So, in 1992, he founded Medispec.

Earlier last decade, his friend's wife suffered from kidney failure with few options. Her illness drove Spector to test his shockwave treatment for CKD. A significant study was conducted through the Mayo Clinic and financed by the National Institutes of Health, showing that what the company thought could theoretically work from animal studies "is more than a reality. The results were above our expectations," Spector said.
Six patients were treated with Curespec's Nephrospec, providing the device's first safety data and the beginnings of its efficacy. In 2021, a patent for the method of this technology was granted by the US patent office.
A multinational Phase I clinical trial is underway in the Philippines and two hospitals in Israel - Shaare Zedek and Sheba Medical Center. The company is expanding the trial into seven more centers - two in the United States and five in Europe.
Because most studies using eHAT have been done in animals, these new clinical trials in human subjects will prove essential for understanding the use and effectiveness of eHAT in the real world.
Spector said the hope is that it could prove effective for using Nephrospec not only for CKD but also for diabetic kidney disease, acute kidney injury, and even kidney transplant. Although there is no research describing eHAT in transplant, the premise is intriguing, he said. Studies are expected to evaluate whether eHAT could improve allograft outcomes in addition to standard immunosuppression in kidney transplants.
"A kidney transplant costs nearly $500,000 to perform," Spector said. "If we could save that money or at least improve the possibility of the transplant to succeed, the effects would be dramatic."
He said, "My dream is to see this technology delay dialysis by at least two to four years for patients." The eHAT is also proving effective at reducing blood pressure and the probability of a stroke, which is often linked to kidney failure.
"When you look at the global market of high blood pressure and CKD, you are talking about a few trillion-dollar market," Spector added. "The dream is first to make this therapy available for the benefit of patients. Secondly, it is to reduce the cost of healthcare."
This article was written in cooperation with Curespec. Learn more.
