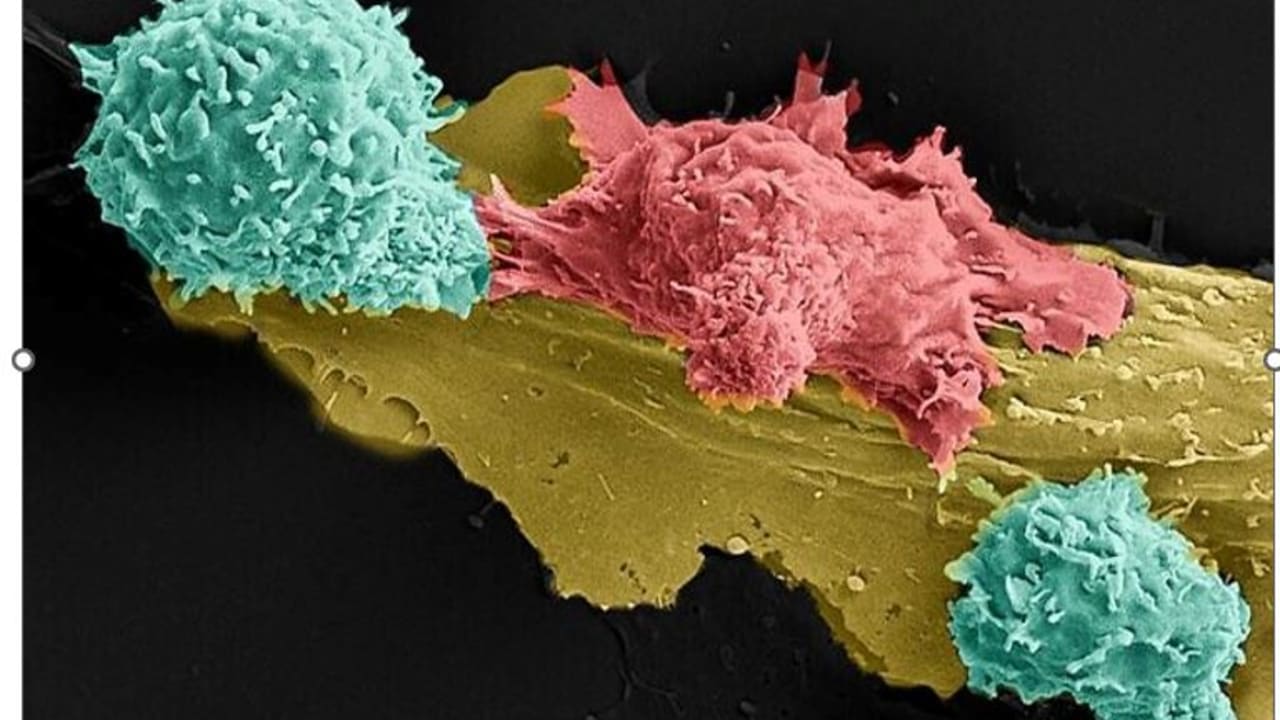
Israeli patients suffering from multiple myeloma (a cancer of white blood cell) or amyloidosis (in which abnormal proteins build up in tissue) have improved or gone into remission with an experimental treatment of CAR-T positive cells.
The medical technology – called HBI0101, anti-BCMA CAR-T cells –was given as part of an ongoing Phase 1b clinical trial at Hadassah-University Medical Center in Jerusalem. Some 85% of the myeloma patients improved, while the complete response rate of the amyloidosis patients reached 100%. In both patient populations, no significant unexpected side effects were observed, the researchers reported.
Multiple myeloma, a cancer of plasma cells that normally produce antibodies, often produces no symptoms initially, but as it progresses, bone pain, anemia, kidney dysfunction and infections may occur. The cause is unknown, but risk factors include obesity, radiation exposure, family history, and certain chemicals. Complications may include amyloidosis, whose vague symptoms include fatigue, peripheral edema, weight loss, shortness of breath, palpitations and feeling faint with standing.
The technology has now been licensed to Immix BioPharma for further development and commercialization. The research, which is at an early phase, was carried out by Hadassah in collaboration with Bar-Ilan University (BIU) in Ramat Gan.
Hadasit, the R&D company of the Hadassah Medical Organization, and BIRAD, its counterpart at BIU, announced that they have entered into the research and license agreement with Immix BioPharma for the development and commercialization of novel tissue-specific therapeutics based on anti-BCMA CAR-T cells targeting plasma cell.

The product of cooperation
This technology has been developed as the result of a collaboration between Prof. Polina Stepensky of the Hadassah-University Medical Center, and Prof. Cyrille Cohen of BIU. Dr. Nathalie Asherie of Hadassah and Ortal Harush of BIU also participated in the research.
Stepensky, head of Hadassah’s department of bone marrow transplantation and cancer immunotherapy, and her team were responsible for the selection of B-cell maturation antigen (BCMA) as the optimal target for the treatment of multiple myeloma and are evaluating this novel CAR-T technology in the clinic.
“We are very excited with the outstanding interim results of our ongoing Phase 1b study in multiple myeloma and amyloidosis AL patients. The licensing agreement will enable us to continue to advance the clinical studies. Our new adoptive cell transfer technology could become one of the first CAR-T treatments for these indications and other BCMA-positive malignancies and has the potential to improve the lives of so many patients globally,” she said.
Stepensky joined forces with Cohen, head of the tumor immunology and immunotherapy lab at BIU and a renowned scientist in the field of immune cells engineering who led the research to built the optimal anti-BCMA CAR construct with proprietary design adaptations. They demonstrated the biological activity of CAR-T cells in the first preclinical studies, leading to establishment of the mutual invention of the researchers.
“For the first time, we successfully reprogrammed a patient’s immune system with a proprietary receptor that we developed in our lab,” he commented. “Through this collaboration with Hadassah, dozens of patients have the future promise of a better treatment option. This successful treatment encourages us to continue developing new approaches for the treatment of cancer and autoimmune diseases.”
“Hadassah has invested millions of dollars of its donors’ money to support this technology during the first crucial stages of development, with the ultimate goal of bringing life-saving CAR-T therapy to cancer patients,” added Prof. Yoram Weiss, director-general of the Hadassah Medical Organization. I would also like to thank the Health Ministry for its support of this project, in recognition of its innovation and importance for Israeli patients. We are delighted that we are able to share this technology at this point in time with the international community.”
BIU chief executive officer Zohar Yinon said: “This is an example of the variety of research in the fields of health and medicine taking place in our labs and of our eco-system that produces cutting-edge research and initiatives that are ready for commercialization by scientists at the faculties of medicine, life sciences, and engineering, the brain center, and the Institute of Nanotechnology and Advanced Materials.”