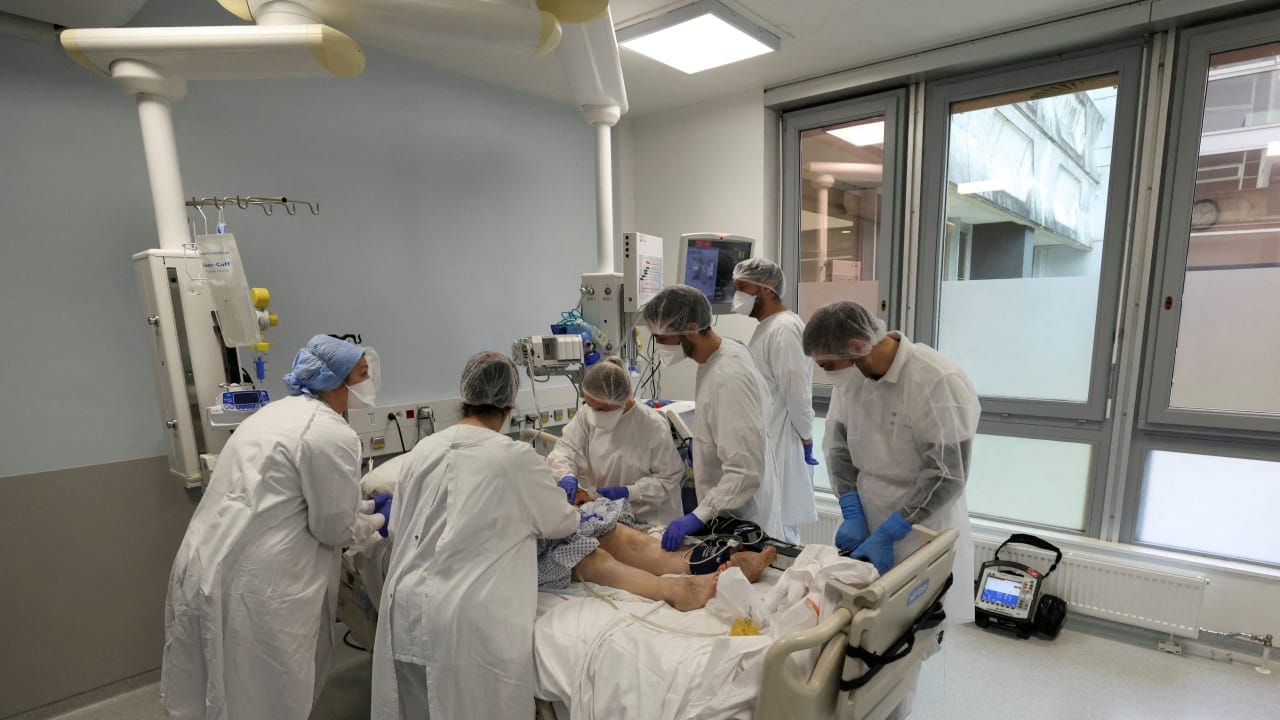
All 18 COVID-19 patients hospitalized with moderate or severe symptoms who received the drug Amor-18 developed by Israeli biotech company Amorphical recovered and were discharged in a few days, the company announced on Wednesday. Of the 19 individuals who were given a placebo, six had to be transferred to intensive care, and two died.
As part of compassionate care, two other patients in a very serious condition were given the drug. Both recovered and were discharged.
“Since the patients treated with the drug recovered within a few days and were released to their home, this was 100% successful,” said Dr. Nashat Abu Saleh, co-director of the coronavirus department at Ziv Medical Center in Safed, where the phase II clinical trial was conducted.
Amor-18, which uses Amorphous Calcium Carbonate (ACC) as the main ingredient, was administered orally or by inhalation. As explained by the company, ACC has the ability to modulate acidic pH changes around each cell. These changes affect the capability of COVID-19 to penetrate the cells and replicate. This allows the drug to prevent the virus from spreading and thus the patients from deteriorating.
“We are excited about the results of the clinical trial, which bring real hope to COVID-19 patients in Israel and around the world, and are especially encouraging these days with the start of the fifth wave of the Omicron variant,” said Amorphical CEO Yossi Ben. “The drug we have developed is anti-inflammatory, safe and effective and it is easy to use.” He noted that Amor-18 will work against the entire SARS virus family, including all COVID variants.
A larger trial is already being conducted at the Ziv, Shamir, Kaplan and Mayanei Hayeshua medical centers in Israel. In the near future, the drug will be tested in other countries. Seven hospitals in Brazil have announced that they will participate, and other centers in Europe and the United States are expected to join soon.