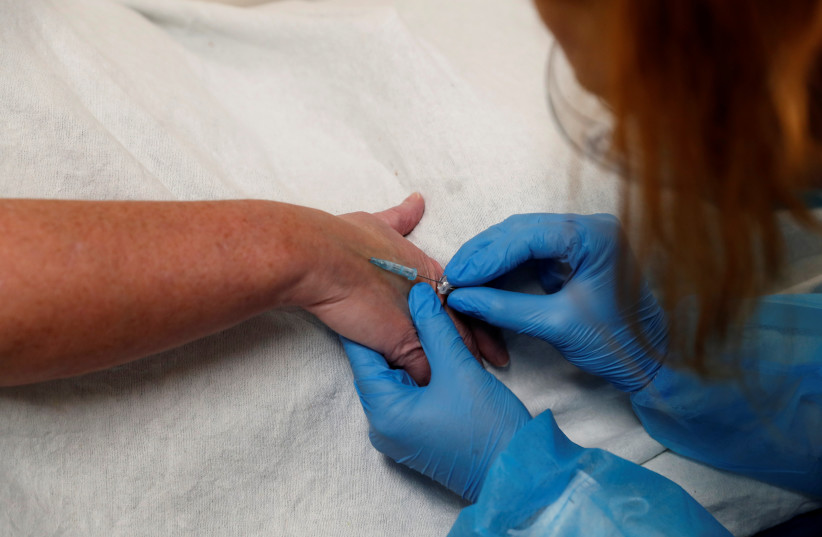
Monoclonal antibody treatments are tailor-made to fight specific infections. In early 2021, the US Food and Drug Administration (FDA) issued emergency authorization for two monoclonal antibody treatments to fight COVID-19. Specifically, they were approved to treat moderate COVID-19 in high-risk, non-hospitalized patients.
During the winter of 2021-2022, the Omicron variant caused a record-breaking surge in COVID-19 infections. The monoclonal antibodies were much less effective against the variant, and thus the FDA unauthorized their use in January 2022. Researchers in Boston, Massachusetts examined the use of monoclonal antibodies after their disauthorization, publishing their findings in the peer-reviewed Journal of the American Medical Association.
“Efforts to improve transparency, equity and value in the COVID-19 response should include public facility-level reporting for all COVID-19 therapies."
Senior author Jennifer Stevens, MD
Researchers at Beth Israel Deaconess Medical Center in Boston examined the US Department of Health and Human Services COVID-19 Reported Patient Impact and Hospital Capacity by Timeseries data set to see if any hospitals continued to prescribe monoclonal antibody treatment after January 2022.
“Continued use of these treatments represents low value care and may reflect conflicting state government guidance or a lack of hospital awareness of deauthorization,” said lead author Timothy Anderson, MD, MAS, Lead for Improving Value in Healthcare at Center for Healthcare Delivery Science at BIDMC and assistant professor of medicine at Harvard Medical School.
“Though the FDA clearly stated these treatments were no longer authorized for use, the FDA did not fully revoke their emergency use authorizations based on the possibility that they may work to treat future COVID-19 variants. This could have led to confusion and misinterpretation.”
What are the numbers?
Researchers discovered that in early 2022, hospitals administered 158,000 doses of unauthorized monoclonal antibody treatments. The use steadily declined overall, although it did vary wildly by state. Florida and New York accounted for a cumulative 44% of monoclonal antibody treatments in 2022. 11 states used more than half of their remaining supply post-deauthorization and 14 used less than 10 percent of it.
“We believe these findings are quite surprising and indicate a need for the FDA to investigate the continued use of treatments found to not be effective for COVID-19,” said senior author Jennifer Stevens, MD, director of the Center for Healthcare Delivery Science at BIDMC and associate professor of medicine at Harvard Medical School.
“Efforts to improve transparency, equity and value in the COVID-19 response should include public facility-level reporting for all COVID-19 therapies. We hope that our findings will lead to greater attention and more diligent regulation by health care providers and government agencies to prohibit the use of unauthorized treatments.”
